目前商用核電站主要采用輕水核反應堆,約占世界核反應堆總數的85%,分為沸水堆和壓水堆2種.此外,重水堆占8%.因此,90%以上的核反應堆的冷卻劑為高溫高壓水.我國核電站目前主要采用壓水堆,在未來20年內,我國核電站的發展仍將以壓水堆為主.
1 高溫高壓水中材料的腐蝕電化學行為
2 高溫高壓水中材料的表面膜特征
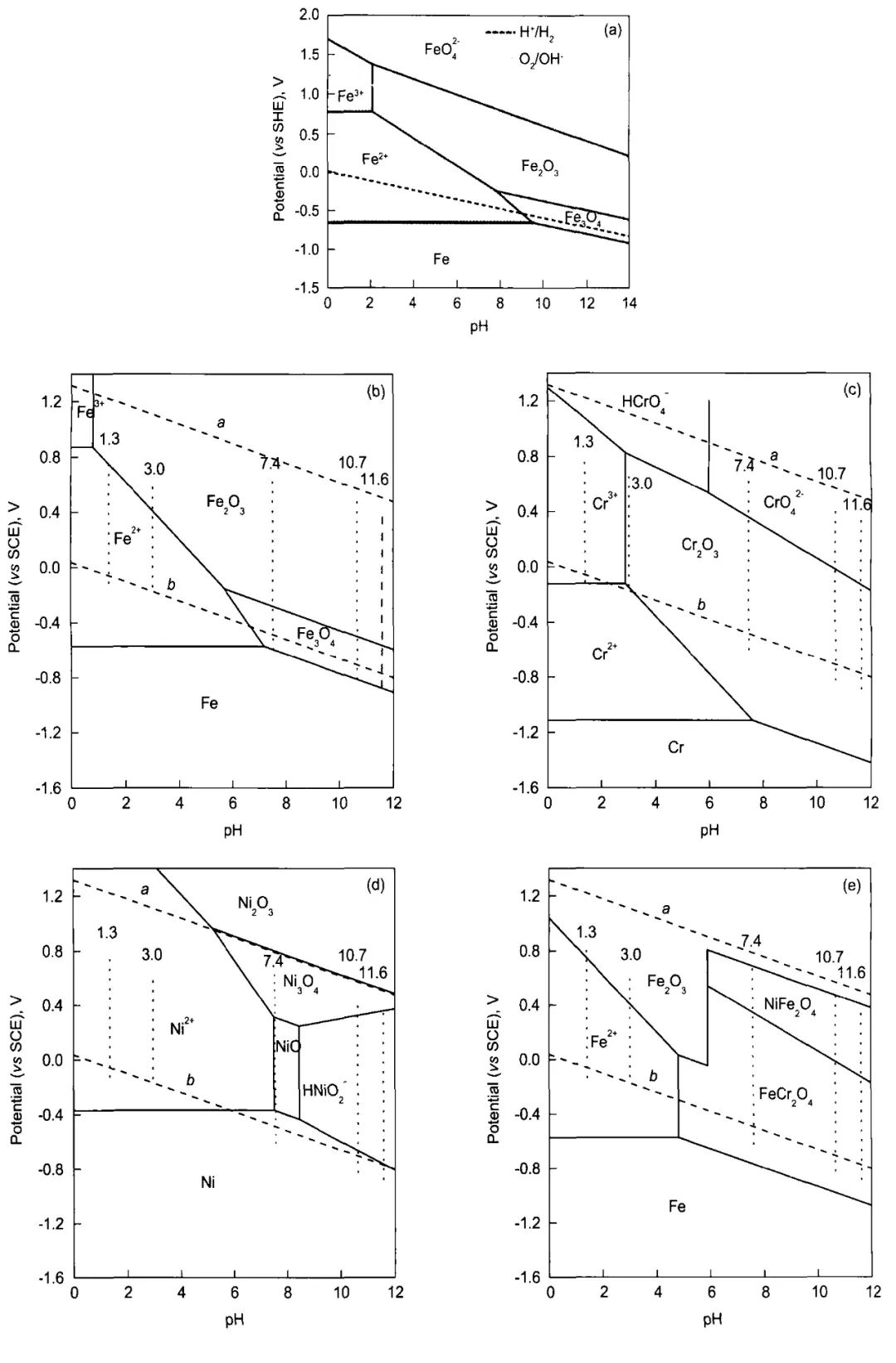
圖1 Fe-H20 25℃[10],Fe-H2O,Cr-H2O,Ni-H2O,Fe-Cr-Ni-H2O 80℃[11]的電位-pH圖
Fig.1 Potential-pH diagrams of Fe-H2O system at 25℃[10](a),Fe-H2O (b),Cr—H2O (c),Ni—H2O (d) and Fe-Cr-Ni-H2O (e) system at 80℃[11](the oxygen and hydrogen equilibria at 1.01×105 Pa are represented by dash lines a and b,respectively,and the vertical dash dot lines reveal the passive regions in different pH solutions)
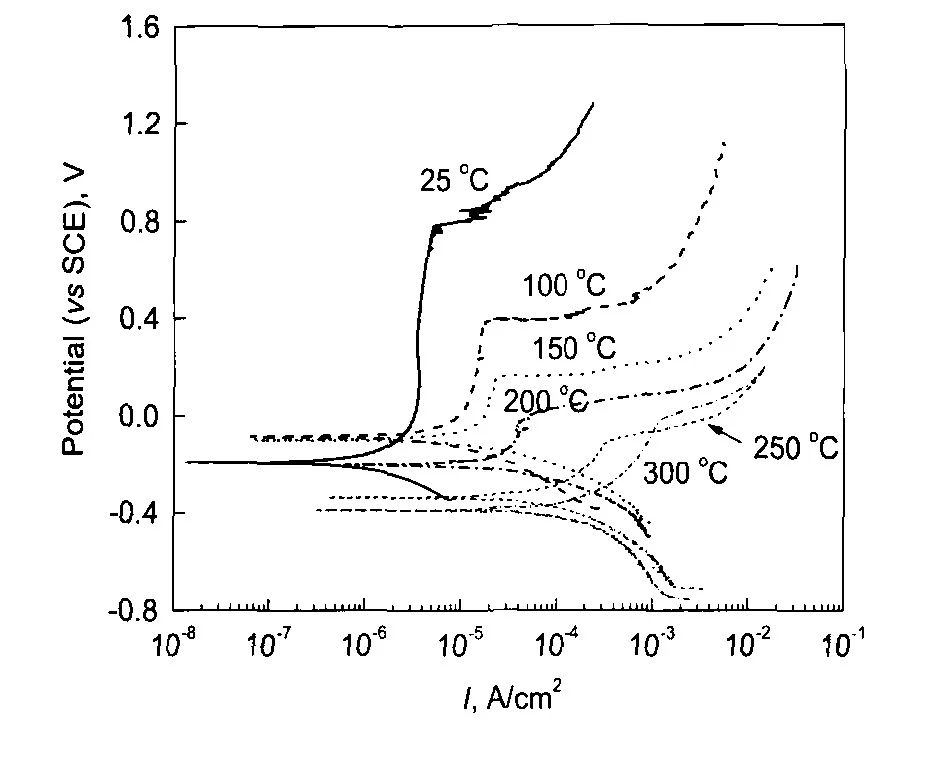
圖2 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中的極化曲線[12]
Fig.2 Polarization curves of 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution consisted of 0.1 mol/L H3BO3+0.0003 mol/L in the temperature range of25—300℃with a scan rate of 1 mV/s[12]
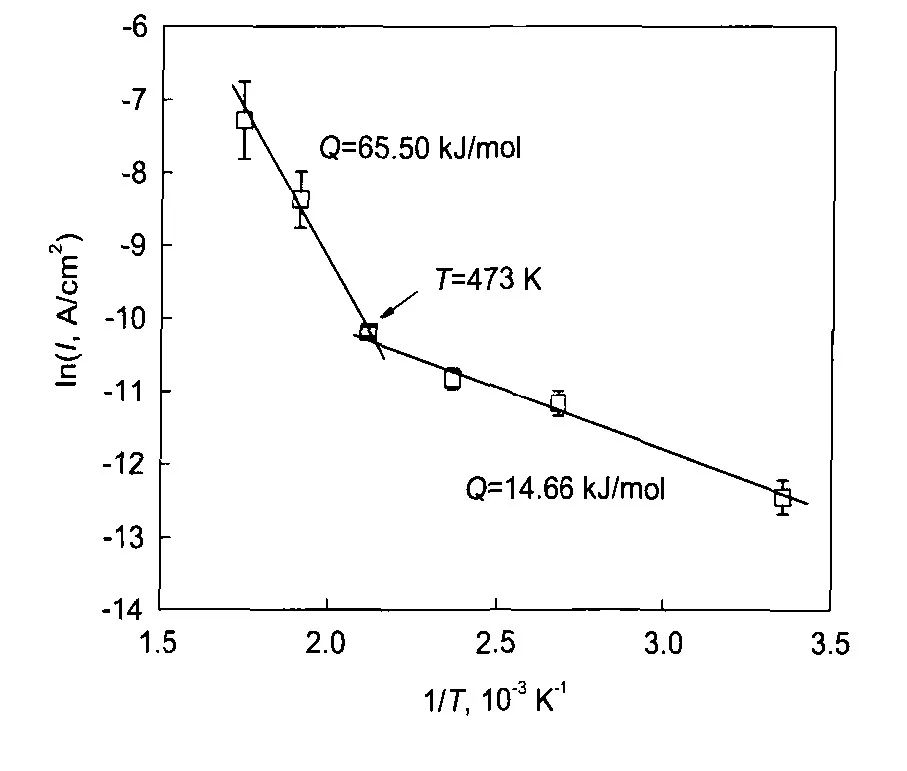
圖3 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中陽極電流密度與溫度的關系[12]
Fig.3 Arrhenius plot of the anodic current density of 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution in the temperature range of 25—300℃[12]
圖4 鎳基合金625在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中的極化曲線[13]
Fig.4 Polarization curves of alloy 625 in 14 MPa and pH(300℃)=6.93 aqueous solution in the temper-ature range of 25—300℃with a scan rate of1 mV/s[13]
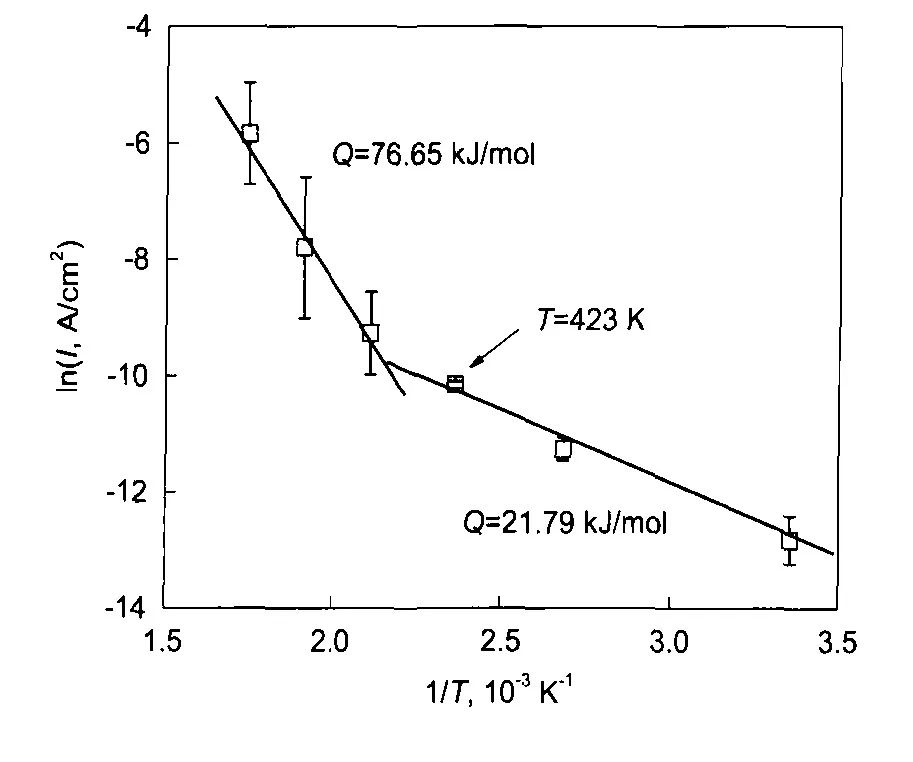
圖5 鎳基合金625在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中的陽極電流密度與溫度的關系[13]
Fig.5 Arrhenius plot of the anodic current density of alloy625 in 14 MPa and pH(300℃)=6.93 aqueous solu-tion in the temperature range of 25—300℃[13]
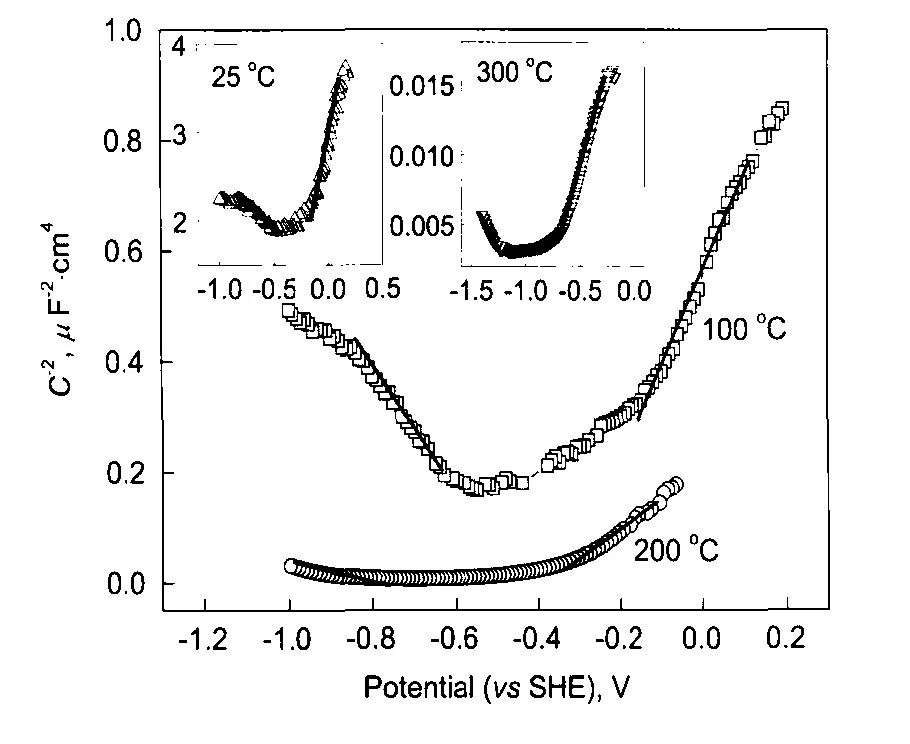
圖6 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜的Mott-Schottky曲線[12]
Fig.6 Mott-Schottky plots of the oxide film on 304SS in14 MPa and pH(300℃)=6.93 aqueous solution in the temperature range of 25—300℃[12]
表1 304不銹鋼在25—300℃和14 MPa以及pH (300℃)=6.93的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜的載流子密度[12]
Table 1 Doping density of the oxide film on 304 stainless steel in 0.1 mol/L H3BO3+0.0003 mol/L LiOH aqueous solution in 14 MPa and pH(300℃)=6.93 in the temperature range of 25—300℃[12]
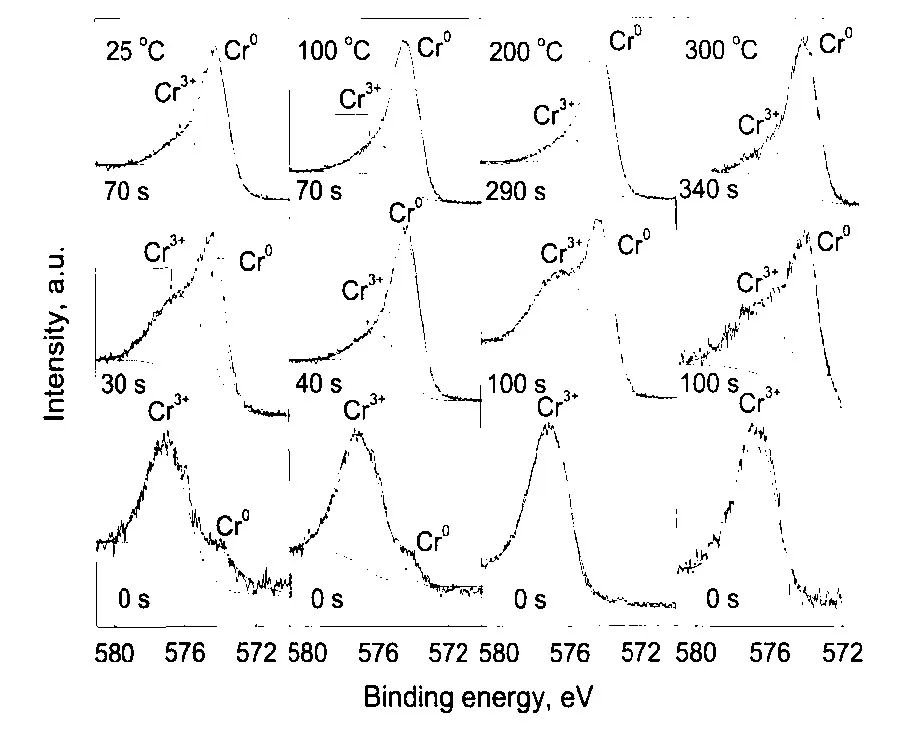
圖7 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜的Cr2p3/2的XPS[12]
Fig.7 Detailed XPS of Cr2p3/2 in the oxide film on 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution in the temperature range of 25-300℃and for different sputtering times[12]
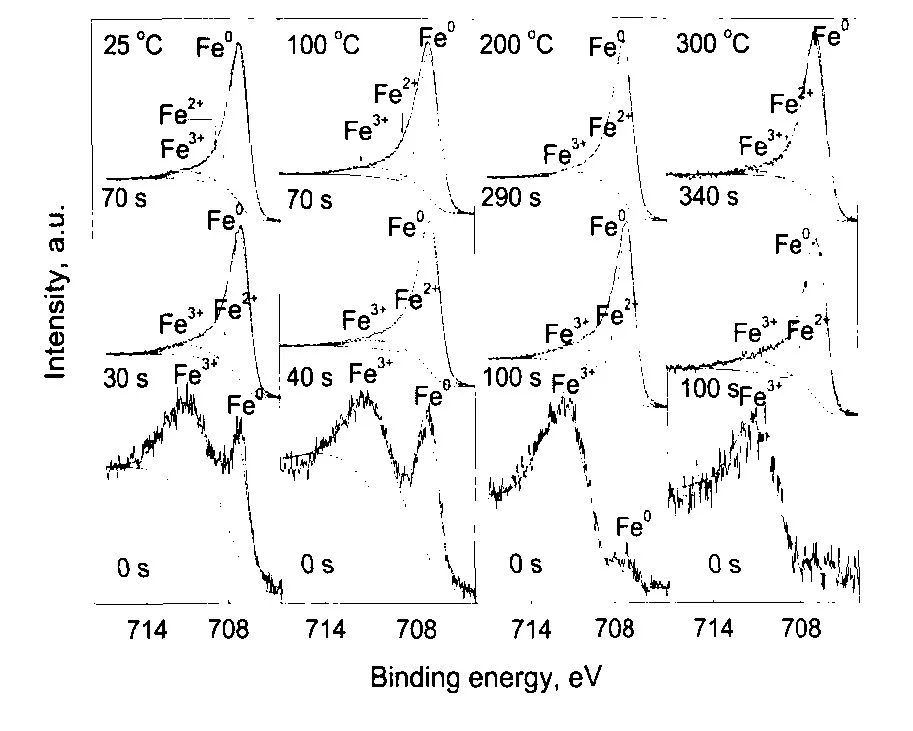
圖8 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜的Fe2p3/2的XPS[12]
Fig.8 Detailed XPS of Fe2p3/2 in the oxide film on 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution in the temperature range of 25—300℃and for different sputtering times[12]
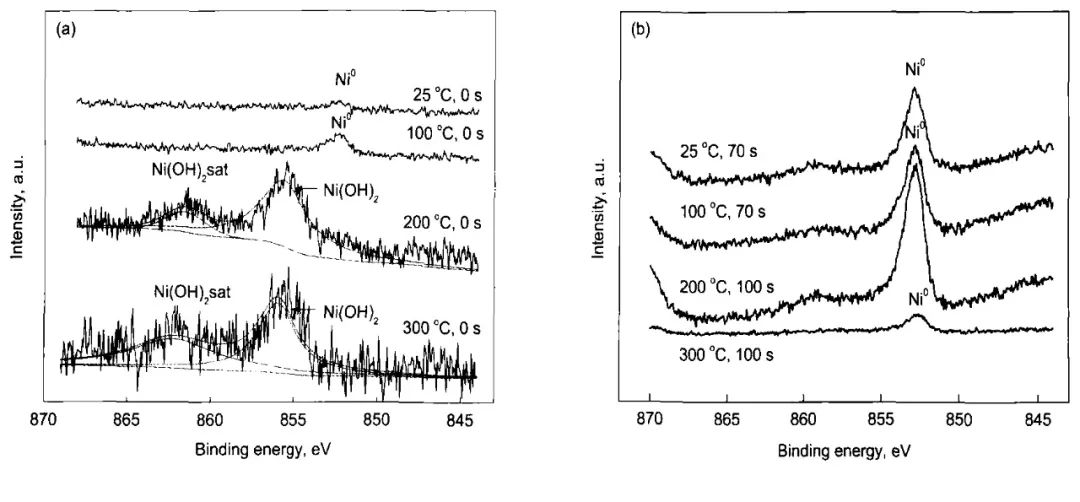
圖9 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中表面及內層生成氧化膜的Ni2p3/2的XPS[12]
Fig.9 Detailed XPS of Ni2p3/2 in the surface (a) and inner layer (b) of oxide film on 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution in the temperature range of 25-300℃and for different sputtering times[12]
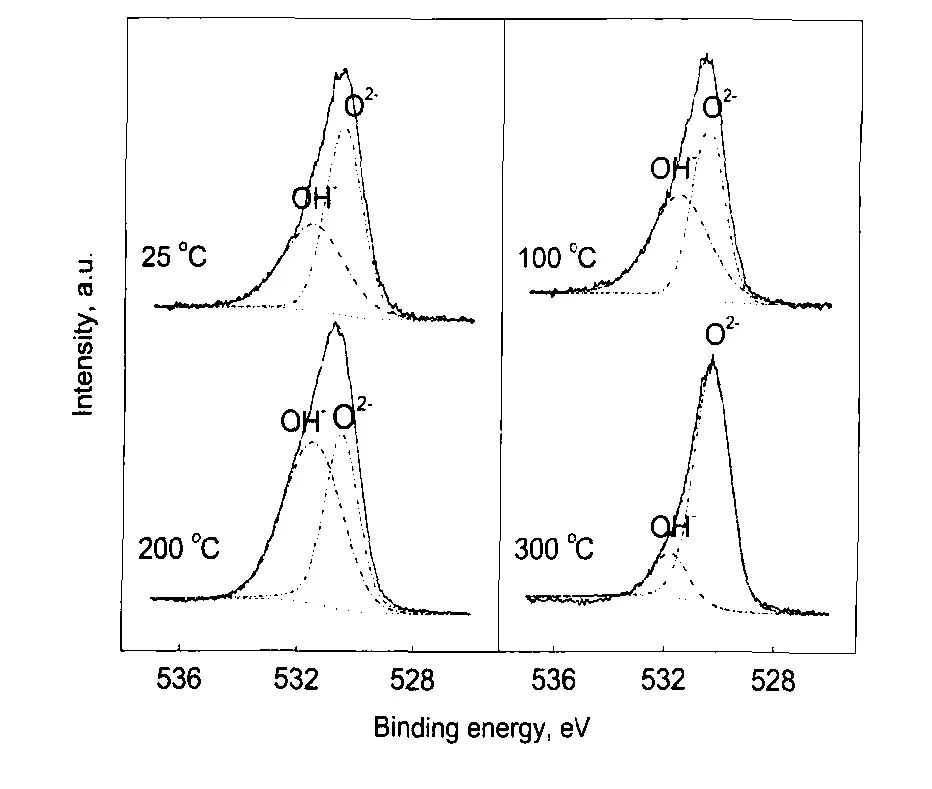
圖10 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜表面的Ols的XPS[12]
Fig.10 Detailed XPS of Ols on the surface of oxide film on304SS in 14 MPa and pH(300℃)=6.93 aqueous so-lution in the temperature range of 25—300℃[12]
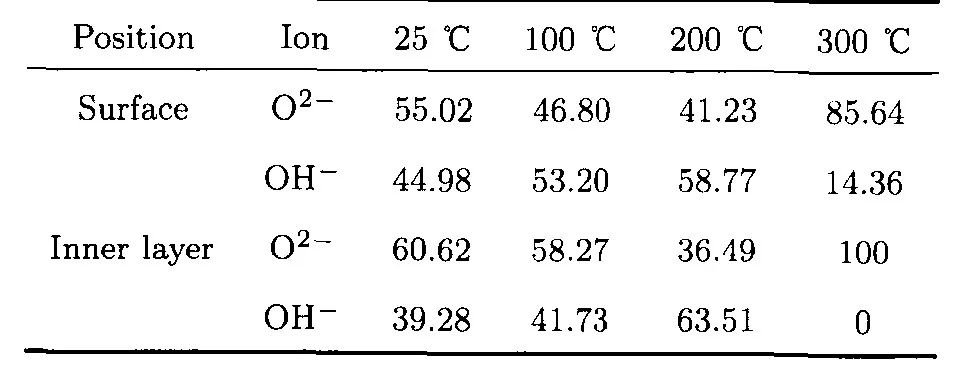
表2 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜表面和內層O2-和OH-在O1s譜中所占比例[12]
Table 2 Contents of O2-and OH-in O1s spectra of the surface and inner layer of the oxide film on 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution and the temperature range of 25—300℃[12]
3 影響材料腐蝕的若干主要因素
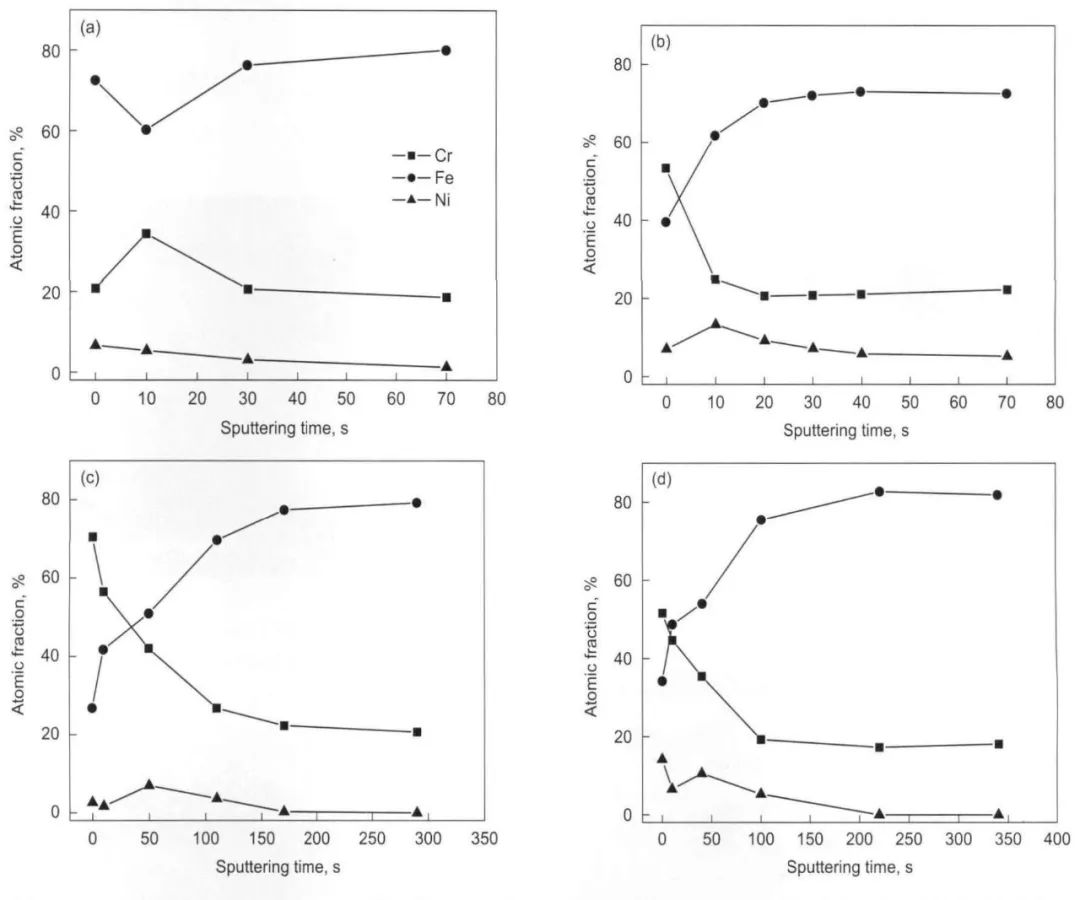
圖11 304不銹鋼在25—300℃和14 MPa的0.1 mol/L H3BO3+0.0003 mol/L LiOH水溶液中生成氧化膜內合金元素的深度分布曲線[12]
Fig.11 Depth profiles of the alloy elements in the oxide film on 304SS in 14 MPa and pH(300℃)=6.93 aqueous solution at 25℃(a),100℃(b),200℃(c) and 300℃(d)[12]
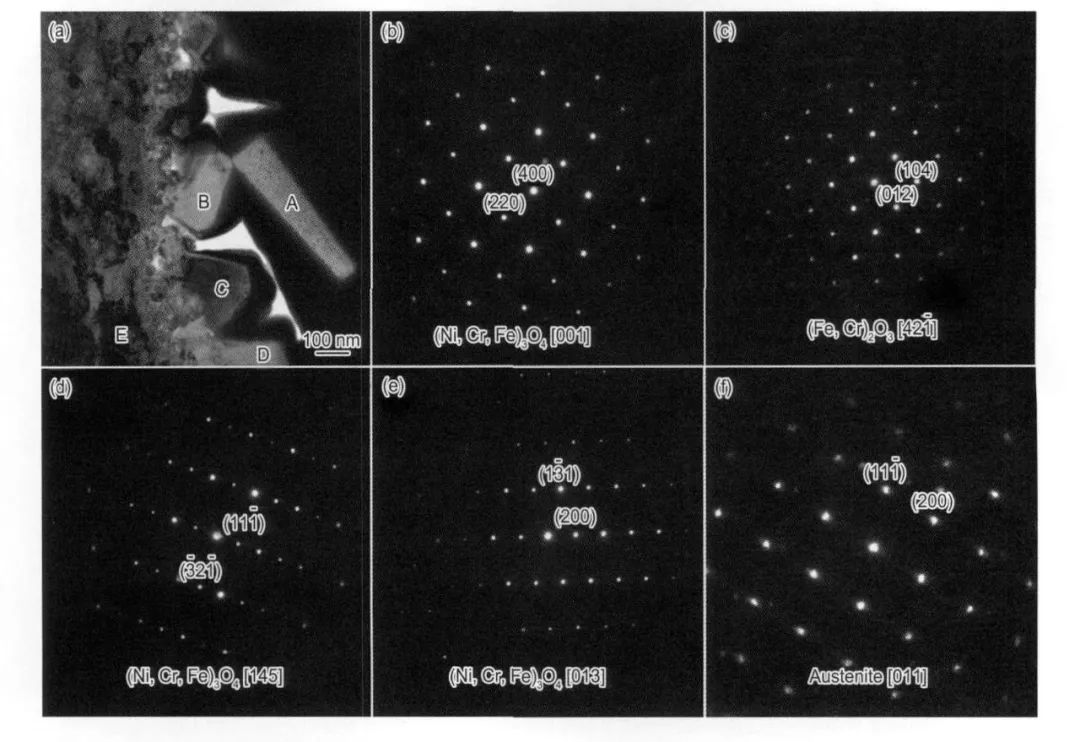
圖12 304不銹鋼在290℃和3×10-6氧的水中實驗290 h后產生的表面氧化膜截面的TEM像和選區電子衍射圖[15]
Fig.12 TEM micrograph (a) and SAED patterns of oxide film corresponding to A (b),B (c),C (d),D (e) and E(f) marked in Fig.12a formed on type 304 SS exposed for 290 h in 290℃water containing 3×10-6 O2[15]
圖13 304不銹鋼在290℃和3×10-6氧的水中產生的表面氧化膜內層的TEM像[15]
Fig.13 TEM morphologies (a,b) and corresponding SAED pattern (c) of the inner layer of oxide film formed on type 304 SS in 290℃water containing 3×10-6 O2[15]
圖14 690合金管材中以MgO為核心的TiN夾雜以及腐蝕后的截面照片,表明在核電一回路水中325℃的恒載荷下暴露在外表面的MgO核心優先腐蝕產生的點蝕[23]
Fig.14 Morphology of TiN inclusion with the MgO nucleus in alloy 690TT (a) and SCC initiating at TiN inclusion located at the near surface of the alloy after constant loading test in 325℃water containing 1500×10-6 B,2.3x10-6 Li,and dissolved 2×10-6 oxygen (b)[23]
圖15 690合金管中條帶狀TiN夾雜的形貌及恒載荷暴露在一回路水中在條帶夾雜處優先發生應力腐蝕[23]
Fig.15 TiN-inclusion striitger at the surface of alloy 690TT (a) and SCC initiation at TiN-inclusion stringer after constant loading test in 325℃water with the addition of 1500×10-6 B,2.3×10-6 Li,and dissolved 2×10-6oxygen (b)[23]
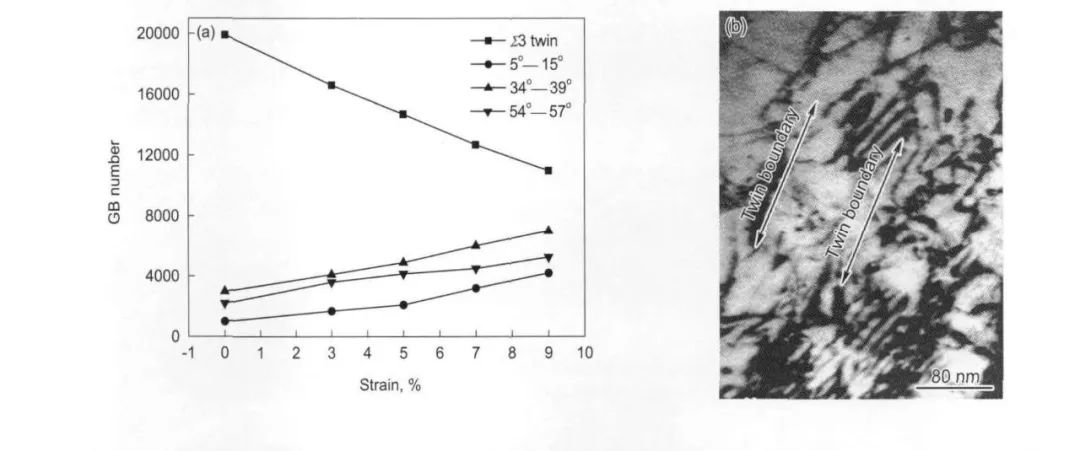
圖16 690合金低能的低∑重位點陣晶界數量隨一維拉伸應變增加而減少,并且在9%拉伸變形后位錯呈塞積結構[24]
Fig.16 The nearly linear relationships between strain amplitude and GB fraction (a) and the TEM observation of dislocation near twin boundaries after X9%tension strain (b)[24]
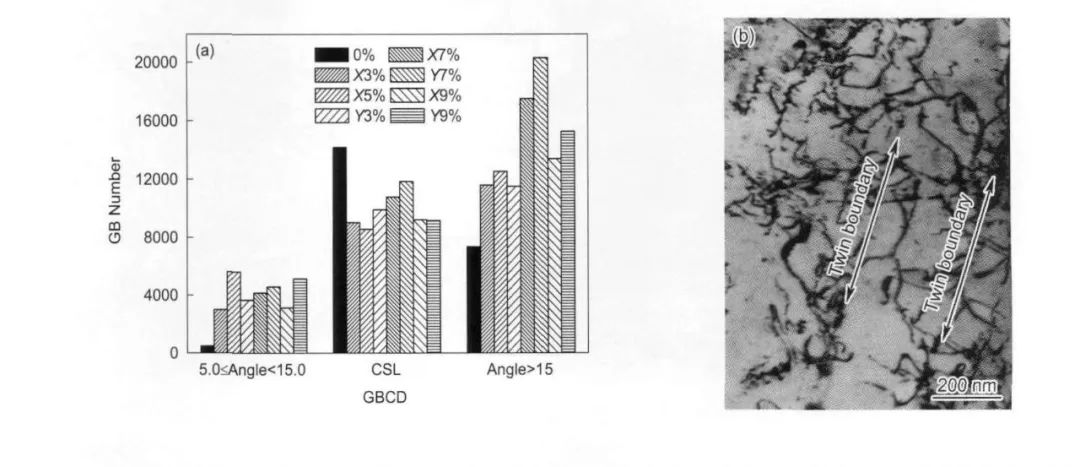
圖17 690合金低能的低∑重位點陣晶界數量隨二維拉伸應變加入出現先減少后恢復,在9%的二維交替拉伸變形后呈位錯纏結結構[24]
Fig.17 GB number distribution of deviation degree from ideal∑3 misorientation with different range of strains (a)and the TEM observation of dislocation near twin boundaries after 2D tension (consecutively X3%,X5%Y3%,X7%,Y7%,X9%and Y9%)(b)[24]
圖18 690合金劃傷的變形和劃傷底部在高溫高壓含Pb堿溶液中的應力腐蝕裂紋萌生[25]
Fig.18 Surface morphologies of the scratch with the size about 50μm in depth and 300μm in width at scratching speed of 9 mm/s[25]
(a) top-view of the scratch (b) the scratch-bank with deformed twin and slip bands (c) the scratch-bed with local “micro-cracks” due to tearing shown as arrows (d) crack initiation at scratch-bank and scratch-bed for alloy 690TT after 30 d immersion in 10%NaOH+10 g/L PbO at 330℃
圖19 690合金劃傷截面形貌、劃傷底部晶粒細化與劃傷的邊緣機械孿晶[26]
Fig.19 Cross-section of the scratch[26]
(a) SEM observation (b) TEM observation of the nanograins at the bottom of the scratch (c) TEM observation of the mechanical twins at the bank of the scratch
圖20 690合金在溶解氧濃度為2×10-6的模擬一回路高溫高壓水中浸泡60 d后,腐蝕產物膜截面的胞狀內氧化膜[27]
Fig.20 Cellular inner oxide film of alloy 690TT after 60 d immersion in simulated primary water with dissolved oxygen content of 2×10-6[27]
4 結論
參考文獻
[1] Macdonald D D,Scott A C,Wentrcek P.J Electrochem Soc,1979;126:1618
[2] Macdonald D D,Liu C,Michael M P.In:Kearns J R, Scully J R,eds.,Electrochemical Noise Measurements on Carbon and Stainless Steel in High Subcritical and Supercritical Aqueous Environments.ASTM STP 1277,1996: 247
[3] Lvov S N,Zhou X Y,Macdonald D D.J Electroanal Chem, 1999;463:146
[4] Zhou X Y,Lvov S N,Wei X J,Benning L G,Macdonald D D.Corros Sci,2002;44:841
[5] Zhang L,Han E H,Ke W,Guan H.Chin Pat,ZL 02132544.8,2005 (張麗,韓恩厚,柯偉,關輝.中國專利,ZL 02132544.8, 2005)
[6] Zhang L,Han E H,Ke W,Guan H.Chin Pat,ZL 02274025.2,2003 (張麗,韓恩厚,柯偉,關輝.中國實用新型專利,ZL 02274025.2,2003)
[7] Guan H,Zhang L,Han E H,Ke W.Chin Pat,ZL 02274026.0,2003 (關輝,張麗,韓恩厚,柯偉.中國實用新型專利,ZL 02274026.0,2003)
[8] Sun H,Wu X Q,Han E H.Chin Pat,CN101470093,2009 (孫華,吳欣強,韓恩厚.中國專利,CN101470093,2009)
[9] Sun H,Wu X Q,Han E H.Chin Pat,CN10147009A.2009 (孫華,吳欣強,韓恩厚.中國專利,CN101470094,2009)
[10] Pourbaix M.Atlas of Electrochemical Equilibria in Aqueous Solutions.Houston:NACE International,1974:121
[11] Huang J B,Wu X Q,Han E H.Corros Sci,2009;51:2976
[12] Sun H,Wu X Q,Han E H.Corros Sci,2009;51:2840
[13] Sun H,Wu X Q,Han E H.Corros Sci,2009;51:2565
[14] Sun M C,Wu X Q,Zhang Z E,Han E H.Corros Sci,2009; 51:1069
[15] Kuang W J,Han E H,Wu X Q,Rao J C.Corros Sci, 2010;52:3654
[16] Sun M C,Wu X Q,Han E H,Rao J C.Scr Mater,2009; 61:996
[17] Sun M C,Wu X Q,Zhang Z E,Han E H.J Supercrit Fluids,2008;47:309
[18] Gao X,Wu X Q,Guan H,Han E H.J Supercrit Fluids, 2007;42:157
[19] Morrison S R.Electrochemistry at Semiconductors and Oxidized Metal Electrodes.New York:Plenum Press, 1980:113
[20] Macdonald D D.J Electrochem Soc,1992;139:3434
[21] Hakiki N E,Montemor M F,Ferreira M G S,Belo M D. Corros Sci,2000;42:687
[22] Staehle R W,Gorman J A.Corrosion,2003;59:931
[23] Meng F J,Wang J Q,Han E H,Ke W.Corros Sci,2010; 52:927
[24] Hou J,Wang J Q,Han E H,Ke W.Mater Sci Eng,2009; A518:19
[25] Meng F J,Wang J Q,Han E H,Ke W.Corros Sci,2009; 51:2761
[26] Meng F J,Han E H,Wang J Q,Zhang Z M,Ke W.Electrochem Acta,2010,doi:10.1016/j.electacta.2010.08.028
[27] Wang J Q,Zhang Z M,Han E H,Ke W.6th SG Conference, Toronto,2009
[28] Dan T C,Shoji T,Lu Z P,Sakaguchi K,Wang J Q,Han E H,Ke W.Corros Sci,2010;52:1228
[29] Hou J,Peng Q J,Sakaguchi K,Takeda Y,Kuniya J,Shoji T.Corros Sci,2010;52:1098
免責聲明:本網站所轉載的文字、圖片與視頻資料版權歸原創作者所有,如果涉及侵權,請第一時間聯系本網刪除。

官方微信
《腐蝕與防護網電子期刊》征訂啟事
- 投稿聯系:編輯部
- 電話:010-62316606-806
- 郵箱:fsfhzy666@163.com
- 腐蝕與防護網官方QQ群:140808414