何松1,邢希金1,劉書杰1,殷啟帥2,耿亞楠1,馮桓榰1,趙秋璇2
(1.中海油研究總院有限責(zé)任公司,北京 100028;2.中國(guó)石油大學(xué) 北京 102249)
摘 要:目的 通過(guò)失重法測(cè)定L80、N80、1Cr、3Cr、9Cr、13Cr等油井管材質(zhì)在硫化氫分壓為0.001、0.01、0.1、0.5、1.26、2 MPa環(huán)境條件下的腐蝕速率。方法 采用高溫高壓反應(yīng)釜對(duì)L80、N80、1Cr、3Cr、9Cr、13Cr等材料在模擬工況下的腐蝕行為進(jìn)行研究。用掃描電子顯微鏡對(duì)所得樣品的腐蝕產(chǎn)物種類、微觀形貌進(jìn)行分析。結(jié)果 在硫化氫分壓為2 MPa以下時(shí),各種材料的腐蝕速率均低于0.125 mm/a,屬于中度腐蝕。而硫化氫分壓為2 MPa時(shí),除9Cr外,其余材料的腐蝕速率均達(dá)到了重度腐蝕以上。不銹鋼的腐蝕速率要明顯低于低合金鋼,且加入少量Cr元素并未對(duì)耐蝕性能有顯著的提升,且某些條件下,腐蝕速率要高于普通低合金鋼。對(duì)于低合金鋼及含Cr量較低的鋼,硫化氫壓力不高于0.1 MPa時(shí),腐蝕速率差異不大,基本保持在0.025 mm/a附近,屬于輕微腐蝕,但當(dāng)硫化氫壓力達(dá)到0.5 MPa時(shí),L80、N80和1Cr的腐蝕速率顯著增高。在硫化氫分壓0.001~0.1 MPa之間,常用油井管材質(zhì)的點(diǎn)蝕嚴(yán)重程度隨硫化氫分壓增大而逐漸增加;在硫化氫分壓0.1~0.5 MPa之間,常用油井管材質(zhì)點(diǎn)蝕程度隨硫化氫分壓增大而逐漸降低;在0.5~2 MPa之間,點(diǎn)蝕程度又逐漸增加。結(jié)論 對(duì)于不銹鋼,當(dāng)硫化氫壓力不高于0.1 MPa時(shí),雖然腐蝕速率隨硫化氫壓力升高,呈現(xiàn)一定的上升趨勢(shì),但腐蝕速率均維持在較低的水平;當(dāng)硫化氫壓力達(dá)到0.5 MPa時(shí),不銹鋼的腐蝕速率顯著增大。不銹鋼的耐蝕性能要遠(yuǎn)優(yōu)于低合金鋼,尤其是在硫化氫壓力較低的環(huán)境中。
關(guān)鍵詞:海上油氣田;管材;防腐;腐蝕速率;均勻腐蝕;點(diǎn)蝕
油套管腐蝕是制約海上油氣田安全鉆井和生產(chǎn)的重要安全因素之一[1-2]。油氣井的溫度、壓力、氣油比、油水比、產(chǎn)水量、酸氣含量及比例,地層水中的Cl-與HCO3-含量,介質(zhì)流速與流動(dòng)狀態(tài),壓力降,凝析液析出量及析出位置,變化的生產(chǎn)條件,封隔器位置,人工井底深度,固相含量,油套管腐蝕失效的類型、程度、分布井段,周邊油氣井的作業(yè)井史及防腐措施等,都是影響和制約油套管腐蝕的因素[3-7]。其中的井下溫度、CO2分壓、H2S分壓、地層水氯離子含量等,是最重要的因素。隨著海上油田開發(fā)的逐步深入,越來(lái)越多的油田和區(qū)塊出現(xiàn)不同程度的硫化氫,嚴(yán)重影響井內(nèi)的油套管安全。目前,國(guó)內(nèi)外學(xué)者通過(guò)室內(nèi)實(shí)驗(yàn)和現(xiàn)場(chǎng)數(shù)據(jù)分析,對(duì)二氧化碳腐蝕機(jī)理和影響因素已經(jīng)有了比較深入的認(rèn)識(shí)[8-14],但是實(shí)驗(yàn)研究用的材質(zhì)普遍較單一。為了更直觀清楚地研究不同材質(zhì)在不同硫化氫分壓下的腐蝕規(guī)律,本文通過(guò)對(duì)L80、N80、1Cr、3Cr、9Cr、13Cr等油套管材料進(jìn)行室內(nèi)實(shí)驗(yàn),提出油套管腐蝕規(guī)律。
1 實(shí)驗(yàn)
實(shí)驗(yàn)溫度為60 ℃,H2S分壓分別為0.001、0.01、0.1、0.5、1.26、2 MPa,腐蝕介質(zhì)為采出水,介質(zhì)流速為 1.5 m/s,實(shí)驗(yàn)材料為 API 5CT 的 L80、N80、1Cr、3Cr、9Cr、13Cr等油套管材料,N80、L80、1Cr三種材料的金相組織均為珠光體和鐵素體,9Cr和13Cr的金相組織為馬氏體,3Cr的金相組織為馬氏體和珠光體。腐蝕速率實(shí)驗(yàn)中使用大連科茂磁力驅(qū)動(dòng)高溫高壓反應(yīng)釜,對(duì) L80、N80、1Cr、3Cr、9Cr、13Cr等材料在模擬工況下的腐蝕行為進(jìn)行研究,高壓釜如圖1所示。

圖1 高溫高壓反應(yīng)釜
腐蝕速率實(shí)驗(yàn)采用失重法,腐蝕介質(zhì)為海上某油田現(xiàn)場(chǎng)采出水。實(shí)驗(yàn)前將采出水靜止,使油水相完全分離,抽取下層水相作為腐蝕介質(zhì)進(jìn)行實(shí)驗(yàn)。將失重試樣機(jī)加工至預(yù)定尺寸后,用耐水砂紙逐級(jí)打磨,對(duì)樣品進(jìn)行打號(hào)標(biāo)記后,放入盛有丙酮的器皿中,用脫脂棉除去試片表面油脂及打磨時(shí)附著的顆粒,再放入無(wú)水乙醇中浸泡約5 min,取出試片放在濾紙上,用冷風(fēng)吹干后,用濾紙將試片包好,貯于干燥器中,放置24 h后,測(cè)量尺寸和稱量,精確至0.1 mg。
將處理后的試片裝在聚四氟乙烯材質(zhì)的夾具上,用聚四氟乙烯螺絲擰緊固定。將裝好試片的夾具固定在高溫高壓釜轉(zhuǎn)軸上,加入腐蝕介質(zhì)使試樣完全浸沒(méi)。將高溫高壓釜密閉,再進(jìn)行試壓,確認(rèn)一切部件正常工作后,打開進(jìn)氣閥門及出氣閥門,使用高純氮?dú)獬? h,以除去安裝過(guò)程進(jìn)入的氧及溶解于腐蝕介質(zhì)中的氧。除氧完畢后,關(guān)閉所有閥門使高壓釜密封,升溫至目標(biāo)溫度后,開啟高溫高壓釜進(jìn)氣閥門,通入H2S氣體達(dá)到目標(biāo)壓力并恒定。通過(guò)高溫高壓釜的磁力驅(qū)動(dòng)帶動(dòng)試片旋轉(zhuǎn),調(diào)節(jié)磁力驅(qū)動(dòng)轉(zhuǎn)速使試片線速度達(dá)到1.5 m/s時(shí),開始計(jì)時(shí)。將高溫高壓釜的溫度維持在目標(biāo)溫度,實(shí)驗(yàn)周期為7天。實(shí)驗(yàn)時(shí)間達(dá)到后,關(guān)閉轉(zhuǎn)動(dòng)電機(jī)及加熱,開啟高溫高壓釜出氣閥門將氣體排到堿液槽,將氣體中和吸收,然后開啟釜蓋,取出試片并進(jìn)行觀察,記錄表面腐蝕及腐蝕產(chǎn)物粘附情況,之后立即用清水沖洗掉殘余腐蝕介質(zhì)并用濾紙吸干,再放入丙酮和酒精中除油除水,冷風(fēng)吹干后拍照、進(jìn)行SEM和EDS分析。如不存在點(diǎn)蝕,則分析均勻腐蝕速率;如存在點(diǎn)蝕,分析點(diǎn)蝕坑形貌、點(diǎn)蝕速率和均勻腐蝕速率。點(diǎn)蝕嚴(yán)重程度按照NACE RP 0775標(biāo)準(zhǔn)中失重及點(diǎn)蝕腐蝕程度分類來(lái)判斷。將試片放入配制好的酸洗液中浸泡5 min,同時(shí)用尼龍刷涮洗試片表面的腐蝕產(chǎn)物。用自來(lái)水沖去表面殘酸后,放入無(wú)水乙醇中浸泡約5 min,清洗脫水后,冷風(fēng)吹干,然后用濾紙將試片包好,貯于干燥器中,放置24 h后稱量,精確至0.1 mg。
2 結(jié)果與分析
2.1 均勻腐蝕速率分析
圖2為不同硫化氫壓力時(shí),碳鋼、低合金鋼和不銹鋼等各種材料的腐蝕速率。從圖中可以看出,各種材料的腐蝕速率在硫化氫分壓為2 MPa以下時(shí),均低于0.125 mm/a,屬于中度腐蝕。不銹鋼的腐蝕速率要明顯低于低合金鋼,且加入少量Cr元素并未對(duì)耐蝕性能有顯著的提升,但在某些條件下,腐蝕速率要高于普通低合金鋼。對(duì)于低合金鋼,硫化氫壓力不高于0.1 MPa時(shí),腐蝕速率差異不大,基本保持在 0.025 mm/a附近,屬于輕微腐蝕;但當(dāng)硫化氫壓力達(dá)到0.5 MPa時(shí),L80、N80和1Cr的腐蝕速率顯著增高;而硫化氫分壓為2 MPa時(shí),除9Cr外,其余材料的腐蝕速率均達(dá)到了重度腐蝕以上。
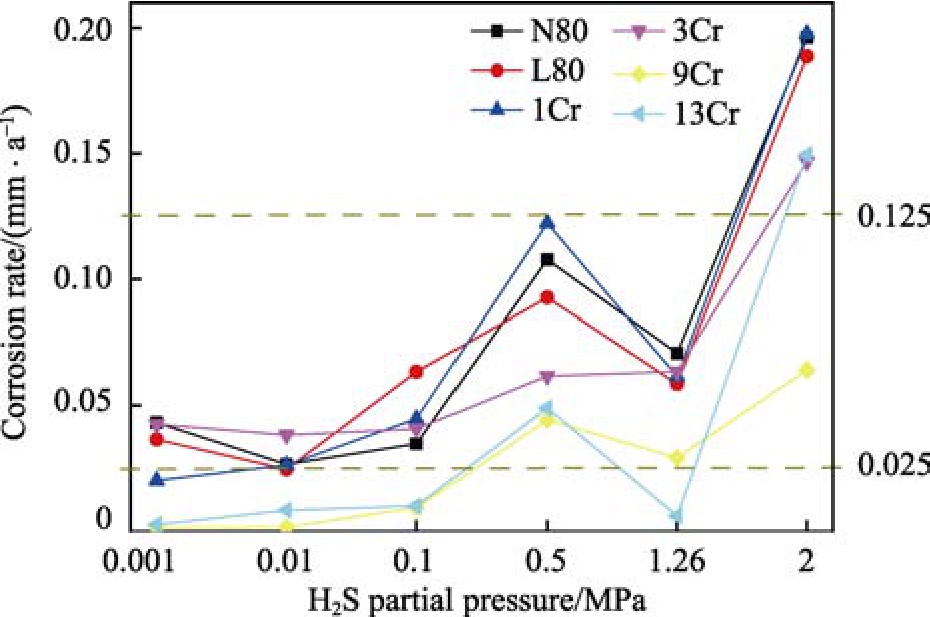
圖2 不同硫化氫壓力時(shí)各種材料的腐蝕速率
相對(duì)而言,3Cr的腐蝕速率增大幅度較小。對(duì)于四種不銹鋼,當(dāng)硫化氫壓力不高于0.1 MPa時(shí),雖然腐蝕速率隨硫化氫壓力升高,呈現(xiàn)一定的上升趨勢(shì),但均維持在較低的水平;當(dāng)硫化氫壓力達(dá)到0.5 Mpa時(shí),不銹鋼的腐蝕速率顯著增大,原因可能是,在高硫化氫分壓下,不銹鋼鈍化膜性質(zhì)發(fā)生了轉(zhuǎn)變,由氧化物轉(zhuǎn)變?yōu)榱蚧铮瑢?duì)金屬基體的保護(hù)能力降低。其中,9Cr和13Cr在0.5 MPa硫化氫條件下的腐蝕速率高于0.025 mm/a。整體來(lái)說(shuō),不銹鋼的耐蝕性能要遠(yuǎn)優(yōu)于低合金鋼,尤其是在硫化氫壓力較低的環(huán)境中。
2.2 點(diǎn)蝕情況分析
20世紀(jì)90年代中期以來(lái),國(guó)內(nèi)外多位學(xué)者系統(tǒng)地研究了鉻含量對(duì)腐蝕速率的影響力,發(fā)現(xiàn)低鉻鋼腐蝕后,鉻元素會(huì)在腐蝕產(chǎn)物膜中富積,這增強(qiáng)了金屬基體的保護(hù)性,但是在不同硫化氫分壓條件下,將碳鋼、低合金鋼、不銹鋼一起研究和分析的較少[15-18]。表1為不同硫化氫壓力時(shí)各種材料的點(diǎn)蝕情況。從表1可以看出,在不同硫化氫分壓條件下,N80、L80、1Cr、3Cr、9Cr、13Cr等材料的點(diǎn)蝕情況各異。此外,常用油井管材質(zhì)在硫化氫分壓0.001~2 MPa范圍內(nèi)有三個(gè)區(qū)間:0.001~0.1 MPa、0.1~0.5 MPa、0.5~2 MPa。硫化氫分壓在0.1~0.5 MPa區(qū)間,出現(xiàn)了點(diǎn)蝕程度降低的現(xiàn)象。分別選取 N80、L80、1Cr、3Cr、9Cr、13Cr等各類材料在硫化氫分壓為 2、0.1、2、2、0.1、0.5 MPa等典型條件下予以分析。
表1 不同硫化氫壓力時(shí)各種材料的點(diǎn)蝕情況

有研究表明,溫度不高時(shí),N80鋼表面主要被FeCO3緊密覆蓋,阻礙腐蝕過(guò)程的進(jìn)行[1]。從表1可以看出,N80材質(zhì)在硫化氫分壓0.001~2 MPa條件下,未發(fā)現(xiàn)點(diǎn)蝕。在2 MPa H2S條件下,N80腐蝕情況如圖3所示。從圖3可以看出,試樣表面附著了一層腐蝕產(chǎn)物,通過(guò)EDS分析后認(rèn)為試樣表面腐蝕產(chǎn)物主要為FeS。
有學(xué)者認(rèn)為,隨著H2S分壓的增加,與在空氣中相比,L80的抗拉強(qiáng)度和延伸率出現(xiàn)不同程度的降低,表現(xiàn)出了較為強(qiáng)烈的氫脆敏感性,點(diǎn)蝕幾率逐漸增大[4-5]。從表 1可以看出,L80只在硫化氫分壓為0.1 MPa時(shí)發(fā)生點(diǎn)蝕。從圖4可以看出,L80材質(zhì)試樣表面附著了腐蝕產(chǎn)物,為FeS。去除腐蝕產(chǎn)物后,發(fā)現(xiàn)L80材料試樣表面有少量微小的點(diǎn)蝕,點(diǎn)蝕速率為0.18 mm/a,屬于輕度腐蝕。
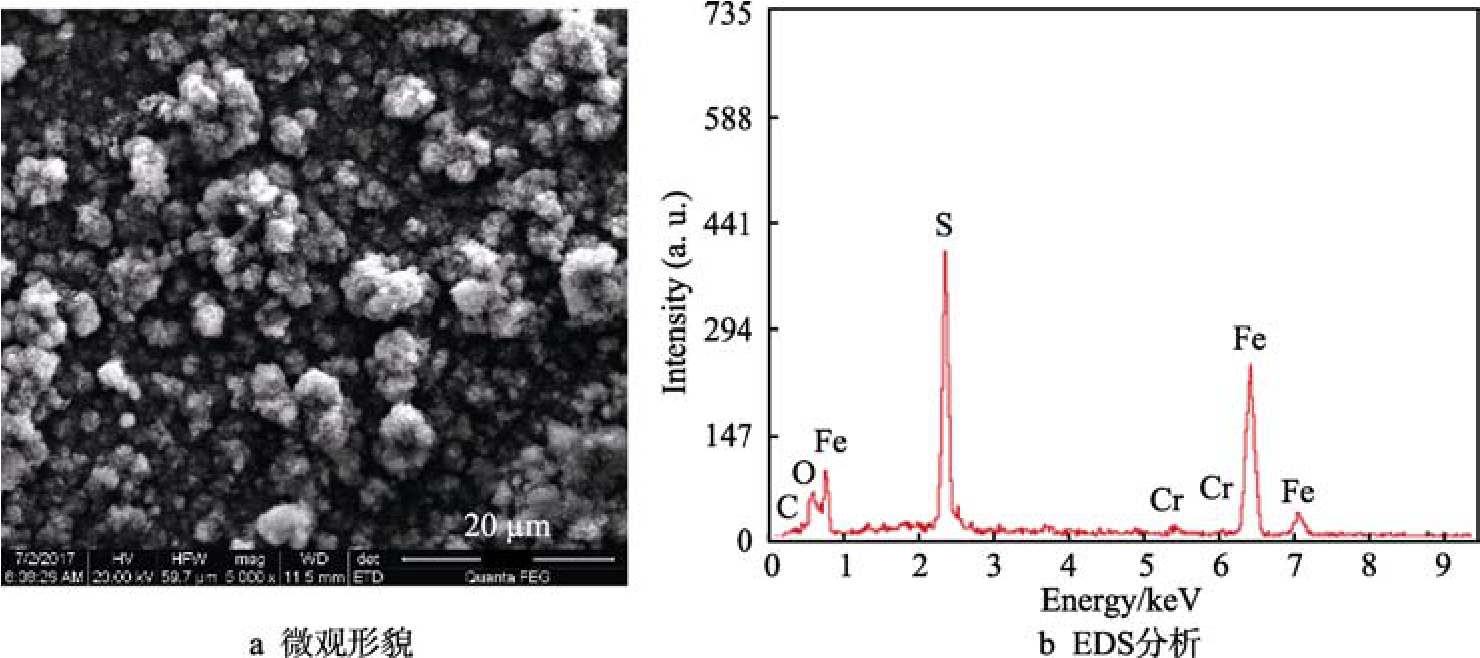
圖3 2 MPa H2S條件下N80的腐蝕產(chǎn)物

圖4 0.1 MPa H2S條件下L80的腐蝕產(chǎn)物
從20世紀(jì)80年代末期開始,國(guó)內(nèi)的鋼材生產(chǎn)企業(yè)寶鋼、天鋼以及阿根廷、日本等國(guó)家相繼開始研究低合金鋼,特別是低含Cr鋼,其成本比碳鋼稍高,但防腐蝕性能大大優(yōu)于普通碳鋼。而在硫化氫條件下,低合金鋼是否能夠抑制點(diǎn)蝕的研究較少[19]。從表1可以看出,1Cr材質(zhì)在硫化氫分壓為0.001~2 MPa的條件下,分別發(fā)生輕度、中度以及無(wú)點(diǎn)蝕等腐蝕現(xiàn)象。從圖5可以看出,在2 MPa H2S條件下,試樣表面附著了腐蝕產(chǎn)物,但成分分析表明主要是鹽的結(jié)晶,而其他試樣表面為腐蝕產(chǎn)物FeS。去除腐蝕產(chǎn)物后發(fā)現(xiàn),1Cr材料試樣表面有少量微小的點(diǎn)蝕,深度較淺,點(diǎn)蝕速率為0.12 mm/a,屬于輕度腐蝕。
從表 1可以看出,3Cr材質(zhì)在硫化氫分壓為0.001~2 MPa的條件下,分別發(fā)生中度、嚴(yán)重以及無(wú)點(diǎn)蝕等腐蝕現(xiàn)象。從圖6的腐蝕產(chǎn)物形貌和EDS分析可以看出,在2 MPa H2S條件下,試樣表面附著了腐蝕產(chǎn)物,但成分分析表明,其主要是鹽的結(jié)晶,而其他試樣表面為腐蝕產(chǎn)物 FeS。去除腐蝕產(chǎn)物后發(fā)現(xiàn),3Cr材料表面有微小的點(diǎn)蝕核,點(diǎn)蝕速率為0.85 mm/a,屬嚴(yán)重點(diǎn)蝕。將1Cr、3Cr材質(zhì)的點(diǎn)蝕情況與L80、N80對(duì)比發(fā)現(xiàn),與二氧化碳條件下的防腐蝕性能不同,在硫化氫環(huán)境條件下,低Cr合金鋼的防點(diǎn)蝕性能低于碳鋼,稍稍優(yōu)于 9Cr、13Cr等含 Cr鋼材質(zhì)。
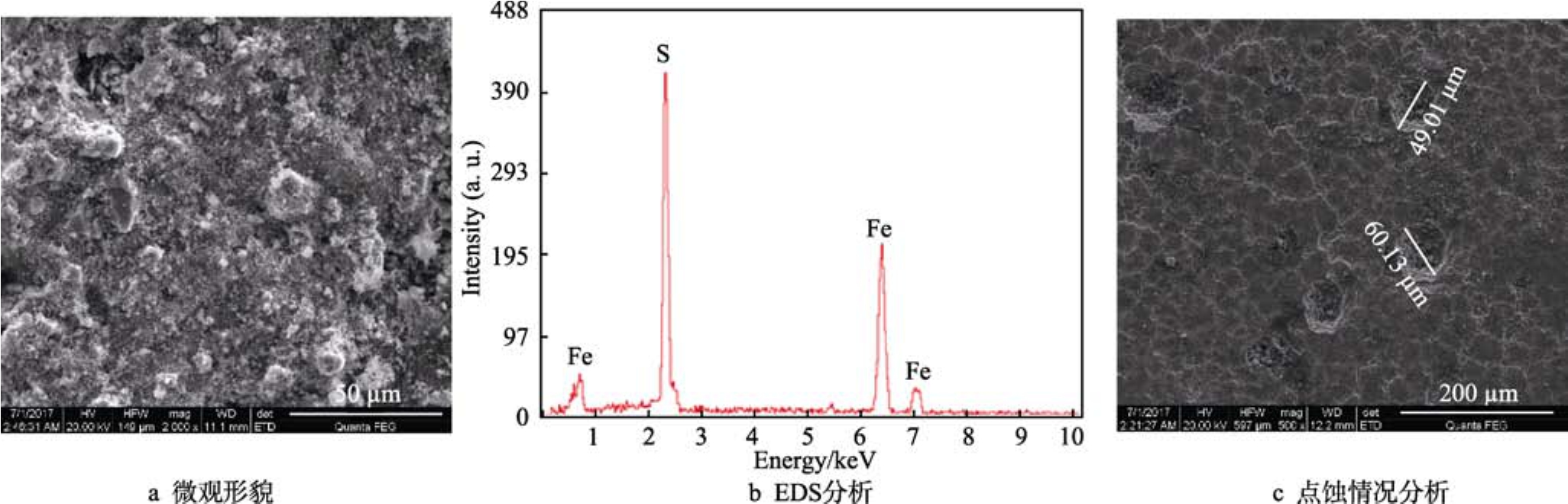
圖5 2 MPa H2S條件下1Cr的腐蝕產(chǎn)物
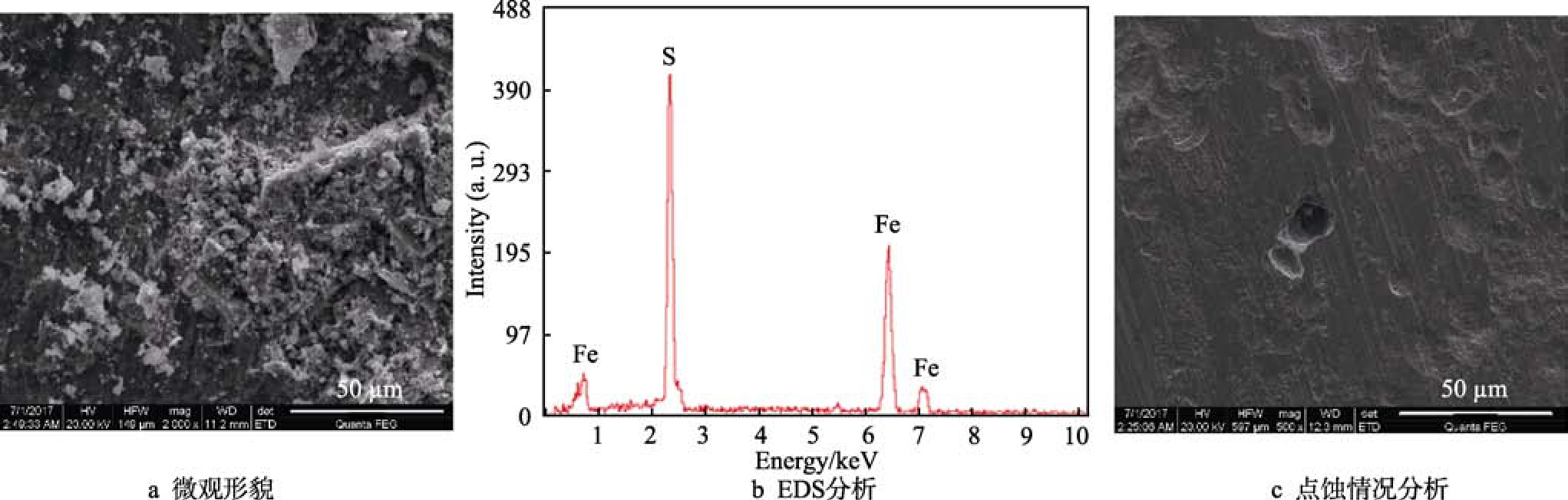
圖6 2 MPa H2S條件下3Cr的腐蝕產(chǎn)物
將 9Cr材質(zhì)的點(diǎn)蝕情況與 1Cr、3Cr材質(zhì)以及L80、N80材質(zhì)進(jìn)行對(duì)比,發(fā)現(xiàn)9Cr材質(zhì)的點(diǎn)蝕情況較為嚴(yán)重。從表1可以看出,9Cr材質(zhì)在硫化氫分壓0.001~2 MPa的條件下,分別發(fā)生中度、嚴(yán)重以及無(wú)點(diǎn)蝕等腐蝕現(xiàn)象。0.1 MPa H2S條件下的腐蝕情況如圖7所示。從圖7的腐蝕產(chǎn)物形貌、EDS分析可以看出,試樣表面附著了腐蝕產(chǎn)物FeS3,其膜很薄,且分布不均勻。去除腐蝕產(chǎn)物后,發(fā)現(xiàn)9Cr材料試樣表面有局部微小的點(diǎn)蝕,點(diǎn)蝕速率為0.56 mm/a,屬于嚴(yán)重腐蝕。
從實(shí)驗(yàn)結(jié)果分析認(rèn)為,13Cr材質(zhì)在硫化氫環(huán)境條件下的防點(diǎn)蝕性能并沒(méi)有隨著含Cr量的增加而優(yōu)于普通碳鋼和低Cr鋼。從表1可以看出,13Cr材質(zhì)在硫化氫分壓0.001~2 MPa的條件下,分別發(fā)生輕度、嚴(yán)重點(diǎn)蝕以及無(wú)點(diǎn)蝕等腐蝕現(xiàn)象。0.5 MPa H2S條件的腐蝕情況如圖8所示。從圖8的腐蝕產(chǎn)物形貌、EDS分析可以看出,試樣表面腐蝕產(chǎn)物很薄,去除腐蝕產(chǎn)物后,發(fā)現(xiàn)13Cr材料試樣表面有明顯的點(diǎn)蝕,點(diǎn)蝕速率為1.87 mm/a,屬于嚴(yán)重腐蝕。
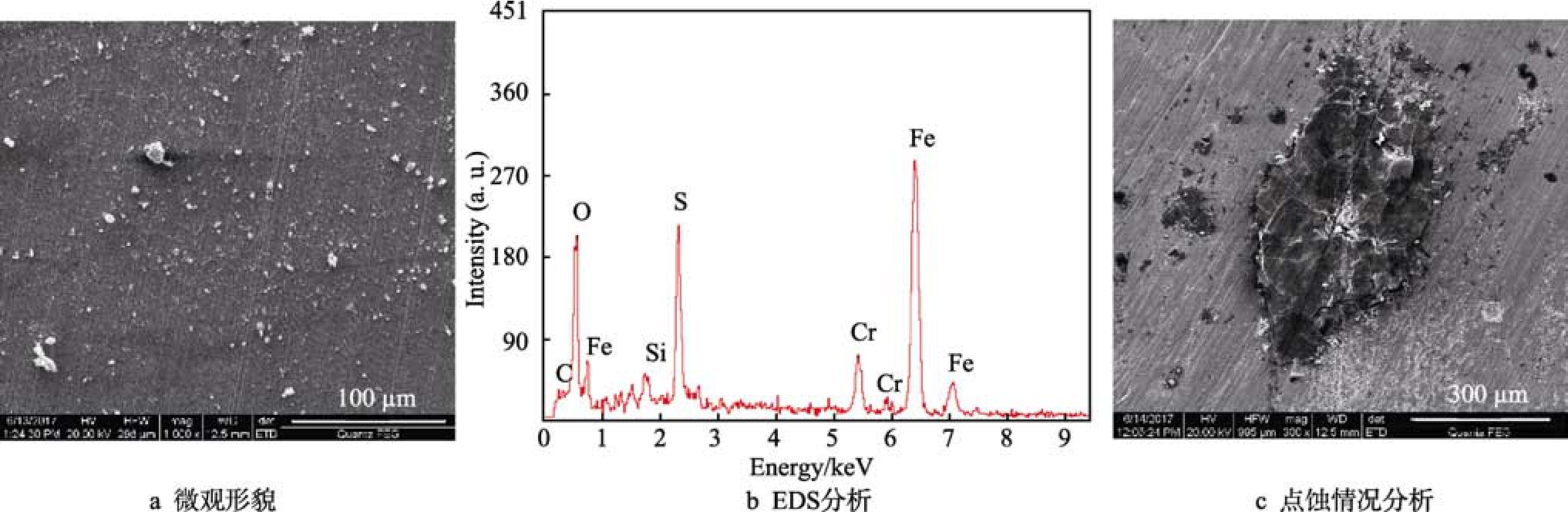
圖7 0.1 MPa H2S條件下9Cr的腐蝕產(chǎn)物
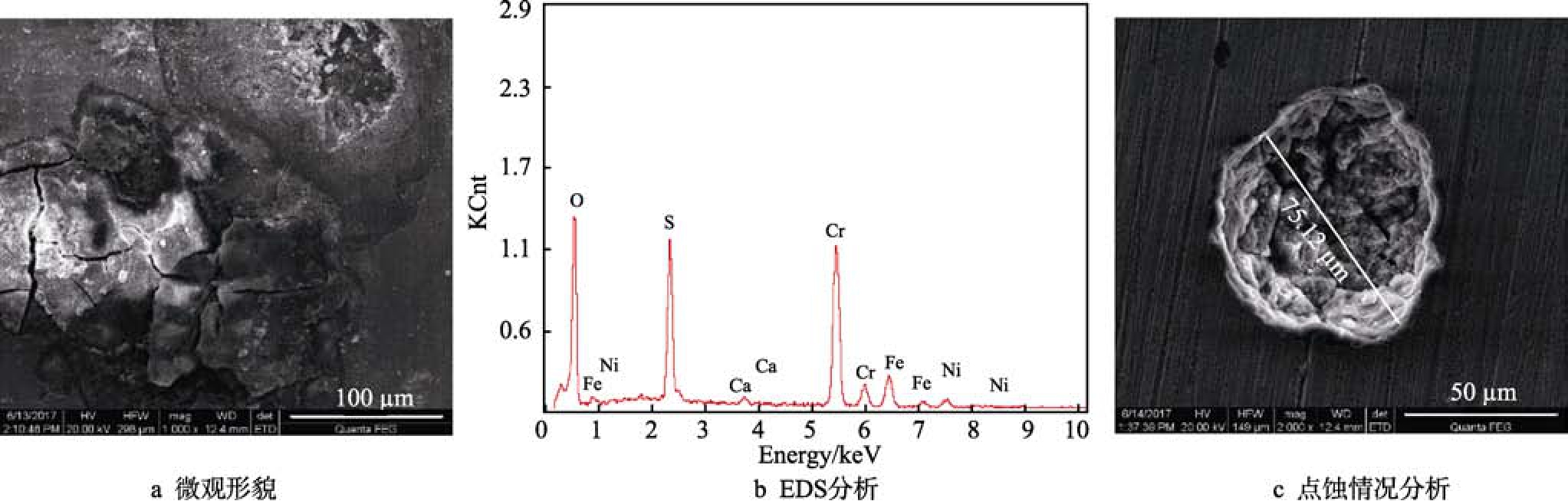
圖8 0.5 MPa H2S條件下13Cr的腐蝕產(chǎn)物
3 結(jié)論
1)從實(shí)驗(yàn)及分析結(jié)果可以看出,在油田工況條件下,隨著硫化氫分壓為 0.001、0.01、0.1、0.5、1.26、2 MPa 逐漸增高,L80、N80、1Cr、3Cr、9Cr、13Cr等材料腐蝕速率均呈增加趨勢(shì),但是硫化氫分壓在1.26 MPa時(shí),油套管材質(zhì)腐蝕速率有下降趨勢(shì)。
2)一般認(rèn)為,Cr含量越高,防腐效果越好,而從本實(shí)驗(yàn)可以看出,在硫化氫防腐中,碳鋼的防點(diǎn)蝕性能在某些條件下有可能優(yōu)于含Cr鋼:N80材質(zhì)在硫化氫分壓為 0.001、0.01、0.1、0.5、1.26、2 MPa時(shí),均未發(fā)生點(diǎn)蝕,其他含Cr鋼材質(zhì)均發(fā)生不同程度點(diǎn)蝕。
參考文獻(xiàn):
[1]謝濤,林海,許杰,等.不同材質(zhì)油套管鋼的 CO2腐蝕行為[J].表面技術(shù),2017,46(1):211-217.XIE Tao,LIN Hai,XU Jie,et al.CO2 corrosion behavior of oil casing steel made from different materials[J].Surface technology,2017,46(1):211-217.
[2]劉陽(yáng),王嘯,許杰,等.渤海凝析油氣藏探井轉(zhuǎn)開發(fā)井防腐技術(shù)研究[J].表面技術(shù),2018,47(10):249-255.LIU Yang,WANG Xiao,XU Jie,et al.Anti-corrosion techniques for transition from exploration well to development well in condensate reservoirs in Bohai sea[J].Surface technology,2018,47(10):249-255.
[3]閆偉,鄧金根,鄧福成,等.油套管力學(xué)-化學(xué)腐蝕規(guī)律分析[J].中國(guó)海上油氣,2014,26(1):87-91.YAN Wei,DENG Jin-gen,DENG Fu-cheng,et al.Discus-sion of mechanical-chemical corrosion in OCTG anti-corrosion design[J].China offshore oil and gas,2014,26(1):87-91.
[4]陳長(zhǎng)風(fēng),路民旭,趙國(guó)仙,等.油套管 CO2腐蝕產(chǎn)物膜的力學(xué)性能[J].金屬學(xué)報(bào),2003,39(2):175-181.CHEN Chang-feng,LU Min-xu,ZHAO Guo-xian,et al.mechanical properties of CO2 corrosion scale on N80 well tube steel[J].Acta metallurgica sinica,2003,39(2):175-181.
[5]張超,張智,曾春珉,等.潿洲-11-4油田含CO2氣井油管柱腐蝕分析[J].中國(guó)海上油氣,2015,27(4):122-125.ZHANG Chao,ZHANG Zhi,ZENG Chun-min,et al.Analysis on tubing corrosion for gas wells with CO2 in WZ 11-4 oilfield[J].China offshore oil and gas,2015,27(4):122-125.
[6]CAI Y D,GUO P C,LIU D M,et al.Comparative study on CO2 orrosion behavior of N80,P110,X52 and 13Cr pipe lines in simulated stratum water[J].Sci China technol sci,2010,53:2342-2349.
[7]VEDAGE H,RAMANARAYANAN T A,MUMFORD J D,et al.Electro-chemical growth of iron sulfide films in H2S-saturated chloride media[J].Corrosion,1993,49(2):114-121.
[8]DURNIE W,MARCO R D,JEFFERSON A,et al.Harmonic analysis of carbon dioxide corrosion[J].Corrosion science,2002,44(6):1213-1221.
[9]TAKABE H,UEDA M.The relationship between CO2 corrosion resistance and corrosion products structure on carbon and low Cr bearing steels[J].Zairyo-to-kankyo,2007,56(11):514-520.
[10]SERRA E,PERUJO A,GLASBRENNER H.Hot-dip aluminium deposit as a permeation barrier for MANET steel[J].Fusion engineering &design,1998,41(1-4):149-155.
[11]董曉煥,趙國(guó)仙,馮耀榮,等.13Cr不銹鋼的CO2腐蝕行為研究[J].石油礦場(chǎng)機(jī)械,2003,32(6):2-3.DONG Xiao-huan,ZHAO Guo-xian,FENG Yao-rong,et al.Study of CO2 corrosion behavior of 13Cr steel[J].Oil field equipment,2003,32(6):2-3.
[12]MISHRA B,ALHASSAN S,OLSON D L,et al.Development of a predictive model for activation-controlled corrosion of steel in solutions containing carbon dioxide[J].Corrosion,1997,53(11):852-859.
[13]WANG C,NEVILLE A.Study of the effect of inhibitors on erosion-corrosion in CO2-saturated condition with sand[J].Spe projects facilities &construction,2009,4(1):1-10.
[14]林玉華,杜榮歸,胡融剛,等.不銹鋼鈍化膜耐蝕性與半導(dǎo)體特性的關(guān)聯(lián)研究[J].物理化學(xué)學(xué)報(bào),2005(7):53-57.LIN Yu-hua,DU Rong-gui,HU Rong-gang,et al.A correlation study of corrosion resistance and semiconductor properties for the electrochemically modified passive film of stainless steel[J].Journal of physical chemistry,2005(7):53-57.
[15]呂詳鴻,趙國(guó)仙,張建兵,等.超級(jí) 13Cr 馬氏體不銹鋼在 CO2及H2S/CO2 環(huán)境中的腐蝕行為[J].北京科技大學(xué)學(xué)報(bào),2010,32(2):207-212.LYU Xiang-hong,ZHAO Guo-xian,ZHANG Jian-bing,et al.Corrosion behaviors of super 13Cr martensitic stainless steel under CO2 and H2S/CO2 environment[J].Journal of Beijing University of Science and Technology,2010,32(2):207-212.
[16]MALKA R,NESIC S,GULINO D A.Erosion-corrosion and synergistic effects in disturbed liquid-particle flow[J].Wear,2007,262:791-799.
[17]UEDA M,TAKABE H.The formation behavior of corrosion protective films of low Cr bearing steels in CO2 environments[J].Annals of surgery,2001,244(5):700-705.
[18]UEDA M,TAKABE H,NICE P I.The development and implementation of a new alloyed steel for oil and gas production wells[J].Free radical biology &medicine,2000,43(3):431-443.
[19]PLACE M C.Corrosion inhibition for severely corrosive gas wells[J].Corrosion,2012,48(4):341-352.
Corrosion Rules of Commonly Used Oil Well Pipes in Hydrogen Sulfide Environment
HE Song1, XING Xi-jin1, LIU Shu-jie1, YIN Qi-shuai2,GENG Ya-nan1, FENG Huan-zhi1,ZHAO Qiu-xuan2
(1.CNOOC Research Institute Limited Liability Company,Beijing 100028,China;2.China University of Petroleum,Beijing 102249,China)
ABSTRACT: The work aims to measure the corrosion rates of L80,N80,1Cr,3Cr,9Cr and 13Cr oil well pipes under the conditions of hydrogen sulfide partial pressure of 0.001,0.01,0.1,0.5,1.26 and 2 MPa by the method of weight loss.The corrosion behavior of L80,N80,1Cr,3Cr,9Cr,13Cr and other materials under simulated conditions was studied by high temperature and high pressure reactor.Scanning electron microscope(SEM)was used to analyze the types and microstructure of corrosion prod-ucts.The corrosion rate of all materials was lower than 0.125 mm/a when the partial pressure of hydrogen sulfide was less than 2 MPa,so the corrosion was moderate.When the partial pressure of hydrogen sulfide was 2 MPa,the corrosion rate of all the materials except 9Cr was higher than that of severe corrosion.The corrosion rate of stainless steel was significantly lower than that of low alloy steel,and addition of a small amount of Cr element could not significantly improve the corrosion resistance.Under certain conditions,the corrosion rate was higher than that of ordinary low alloy steels.For low alloy steel and low Cr steel,the corrosion rate was not much different when the hydrogen sulfide pressure was not higher than 0.1 MPa,and the corrosion rate was basically kept near 0.025 mm/a,so the corrosion was slight.However,when the hydrogen sulfide pressure reached 0.5 MPa,the corrosion rates of L80,N80 and 1Cr increased significantly.The severity of pitting on commonly used oil well pipes gradually increased with the increase of partial pressure of hydrogen sulfide when the partial pressure of hydrogen sulfide was from 0.001~0.1 MPa.When the partial pressure of hydrogen sulfide was 0.1~0.5 MPa,the pitting degree of commonly used oil well pipes decreased with the increase of partial pressure of hydrogen sulfide.The degree of pitting gradually increased again when the partial pressure of hydrogen sulfide was from 0.5~2 MPa.For stainless steel,when hydrogen sulfide pressure is not higher than 0.1 MPa,the corrosion rate of stainless steel increases with the increase of hydrogen sulfide pressure,but the corrosion rate remains at a low level.When the hydrogen sulfide pressure reaches 0.5 MPa,the corrosion rate of stainless steel increases significantly.The corrosion resistance of stainless steel is much better than that of low alloy steel,especially in the environment of low hydrogen sulfide pressure.
KEY WORDS: offshore oil and gas fields;pipes;corrosion protection;corrosion rate;uniform corrosion;pitting corrosion
中圖分類號(hào):TG174.4
文獻(xiàn)標(biāo)識(shí)碼:A
文章編號(hào):1001-3660(2018)12-0014-07
DOI:10.16490/j.cnki.issn.1001-3660.2018.12.003
收稿日期:2018-05-11;
修訂日期:2018-07-13
Received:2018-05-11;
Revised:2018-07-13
基金項(xiàng)目:國(guó)家科技重大專項(xiàng)(2017ZX05032-004);國(guó)家自然科學(xué)基金(51774301,51434009)
Fund:Supported by Major National Science and Technology Projects(2017ZX05032-004),National Natural Science Foundation of China(51774301,51434009)
作者簡(jiǎn)介:何松(1985—),男,碩士,工程師,主要研究方向?yàn)殂@完井、油田化學(xué)。
Biography:HE Song(1985—),Male,Master,Engineer,Research focus:drilling,completion and oilfield chemistry.
通訊作者:耿亞楠(1968—),男,碩士,高級(jí)工程師,主要研究方向?yàn)槭豌@井。郵箱:geyn@cnooc.com.cn
Corresponding author:GENG Ya-nan(1968—),Male,Master,Senior engineer,Research focus:drilling.E-mail:geyn@cnooc.com.cn
免責(zé)聲明:本網(wǎng)站所轉(zhuǎn)載的文字、圖片與視頻資料版權(quán)歸原創(chuàng)作者所有,如果涉及侵權(quán),請(qǐng)第一時(shí)間聯(lián)系本網(wǎng)刪除。

官方微信
《中國(guó)腐蝕與防護(hù)網(wǎng)電子期刊》征訂啟事
- 投稿聯(lián)系:編輯部
- 電話:010-62316606-806
- 郵箱:fsfhzy666@163.com
- 中國(guó)腐蝕與防護(hù)網(wǎng)官方QQ群:140808414





